(EOC Q2) Which of the following amino acid R-groups has a pKa of about 10 Glu O Cys O Lys O Tyr O His 12. For the peptide Ala-Arg-Lys-Ala-Asn-Ser-Ala-Ser, what would be the expected charges at pH 1, 7, and 13 O +3, +3,0 0 +2, +2,-1 0 +3, +2,-1 11. O reducing agents are required to unfold proteins.ġ0. O the protein could be refolded correctly only if the disulfide bonds were left intact. We have observed distinct differences in the frequencies of occurrence of polar and hydrophobic amino acids at positions 1-18 in amphipathic and nonamphipathic helices. O a denatured protein loses its enzymatic activity. We have analyzed the distribution of net charge, helix length, normalized frequency of occurrence, and propensities of the 20 amino acids in the delineated 18-residue helices. Anfinsen involving ribonuclease was that O the information on how the protein should fold was contained in the amino acid sequence. The major conclusion of the experiment of C. O they have polar residues in the N-terminal part and charged residues in the C-terminal part. they have large R-groups on one side and small R-groups on the other as small groups are easier to pack in the interior of the broten o they have one side or face that is predominately polar with the other side being predominately hydrophobic. This means that O they have positive charges on one side and negative charges on the other.
a-Helices and B-sheets are often found to be amphipathic.
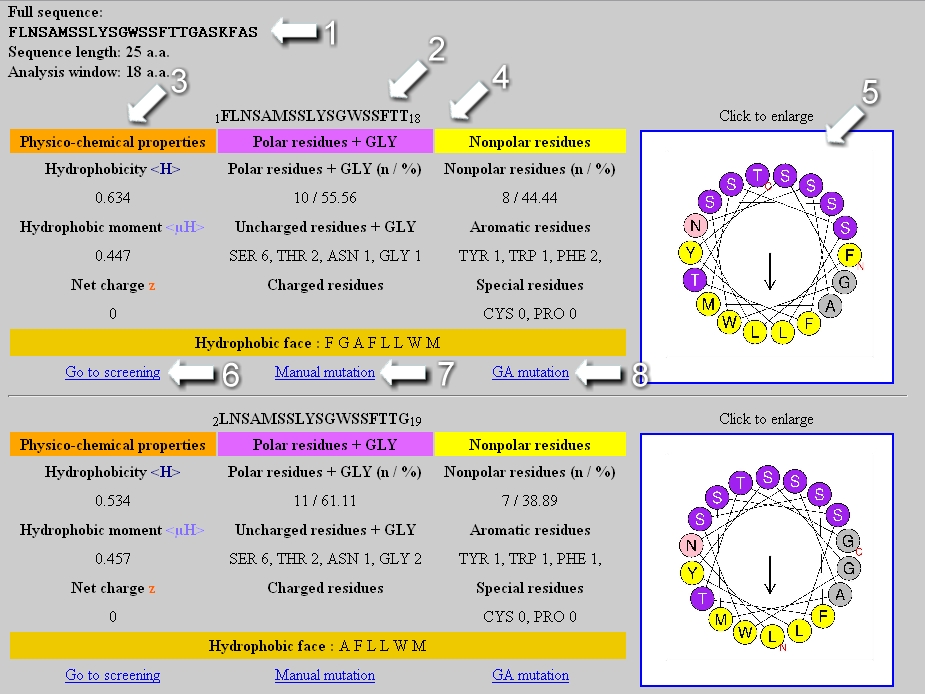
O hydrophilic and hydrophobic amino acid R-groups form hydrogen bonds with each other. hydrophilic amino acid R-groups are on the interior of the protein and hydrophobic groups are on the outside O all peptides form hydrogen bonds with water. Water-soluble proteins such as myoglobin tend to fold such that O hydrophobic amino acid R-groups are on the interior of the protein and hydrophillc groups are on the outside. O Both are stabilized by glycine and proline residues. O The length of a 10-amino acid a-helix and ß-sheet strand will be the same. B Peptide sequences with their N-terminal, by convention, to the left and the hydrophobic residues shaded.T do a-helices and B-sheets have in common? O Both are stabilized by hydrogen bonding involving carbonyl oxygens and amide nitrogens O The same amino acids stabilize both forms of secondary structure. Numbers refer to the amino acid numbers with their side represented by the projected lines. The circle represents the helix viewed from the top with residue 1 closest to the reader. SLIKSVIEMVDEWFRTFL FLIRVLRKVFRVLTRILS RLFRSRVLKIAVIRFLLI A Helix wheel projection. Which of the following sequences can form an amphiphilic helix? (You can use the mnemonic FAMILY VW to remind you which amino acids are hydrophobic).

4a) and determine if the hydrophobic and hydrophilic residues segregate on opposite sides of the helix.

Secondary structure consists of the sequence of amino acids and the intermolecular forces and covalent bonds that form between amino acid backbone components. Primary structure consists of the sequence of amino acids making up the polypeptide. A simple way to decide if a sequence of amino acids can form an amphiphilic helix is to arrange the amino acids around the so called "helix-wheel projection" (Fig. A protein molecule can have four different structural levels. Transcribed image text: Alpha helices are often embedded in a protein so that one side of the helix faces the surface (and is typically hydrophilic) and the other side faces the interior of the protein (and is usually hydrophobic).
